 The ionic lattice ensures that each ion forms regular patterns with the oppositely charged ions surrounding it. These compounds arrange themselves into a “lattice” structure. The electrostatic attraction between the participants facilitates electron transfer between them. This interaction typically occurs between a metal, which carries positive charge, and a nonmetal, which carries negative charge. What are ionic compounds? Ionic compounds form when atoms or molecules attract others with opposite charges. Can stand alone as polyatomic cation and anion.Polyatomic Ionic Compound: each constituent contains atoms from more than one element.Binary Ionic Compound: has two constituents, each of which is comprised of atoms from a single element.Anion participant is almost always a non-metal.Cation participant is almost always a metal.Ionic bond: compounds formed by the transfer of electrons between cations and anions.Anion: positively charged ion formed when neutral atom loses electrons.Cation: positively charged ion formed when neutral atom loses electrons.Charge: found by subtracting (protons-electrons) changes during ionic bonding due to electron transfer.Ion: atom or group of atoms that possess an electric charge. 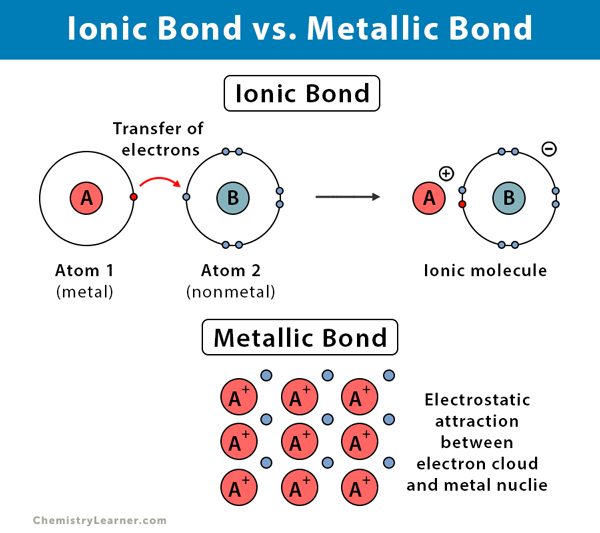
Compound: substance containing molecules that consist of atoms from two or more chemical elements (can be ionic or covalent).We will derive names from the chemical formulas of binary and polyatomic ionic compounds. In this tutorial, you will learn about nomenclature and naming ionic compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
